
Photo credit: Brian Hampson & Irene Chu
Common name: Viola
Latin Origins: becoming violet
Distribution: Nicobar Islands; Phuket, Thailand; Cebu Island, Philippines; several Japanese Islands; Tanzania and Zanzibar. Bangladesh [6]
Habitat: Mangroves
Ecology: Supra littoral; juveniles frequent mangrove forests; adults frequently found on beaches. [1]
Larval Stages: Four larval stages over 15-19 days to reach megalopa [5]
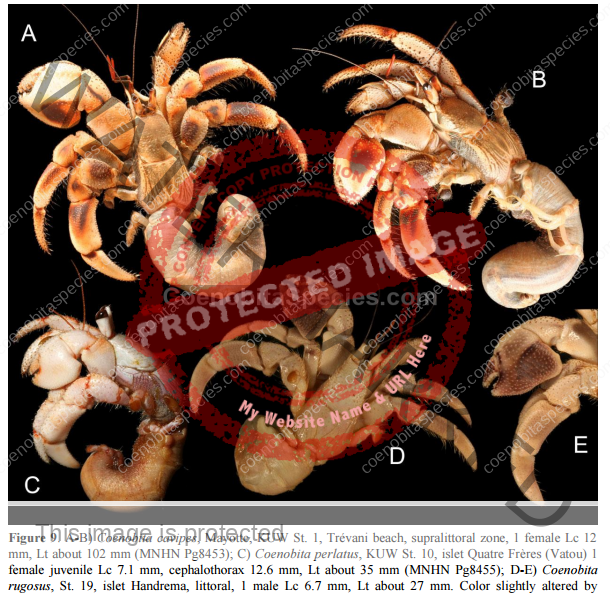
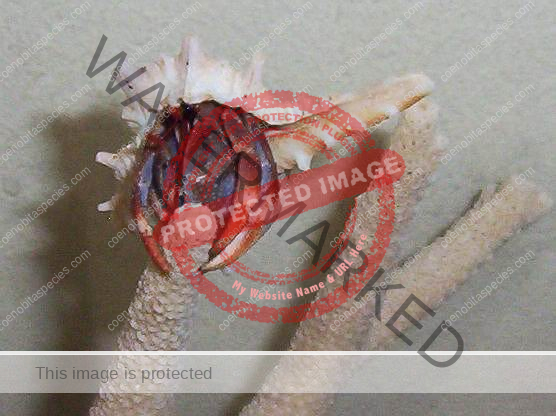
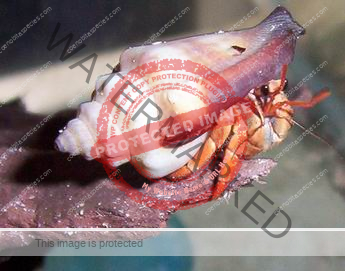
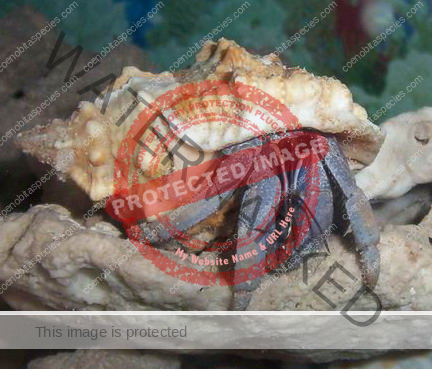
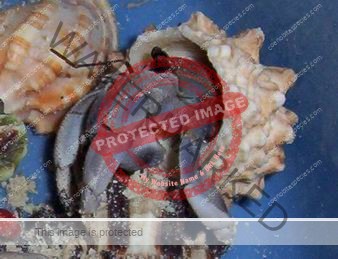
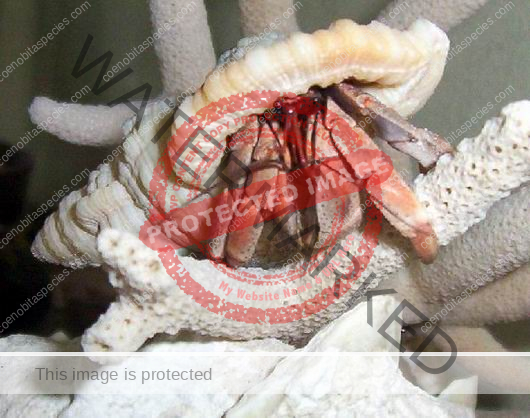
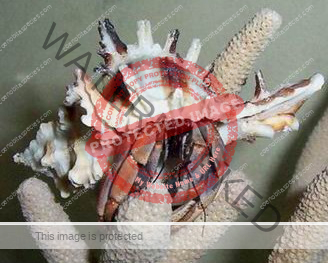
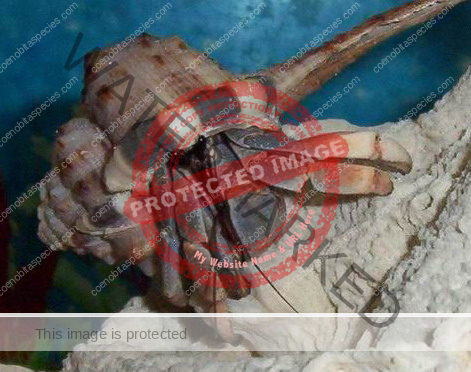
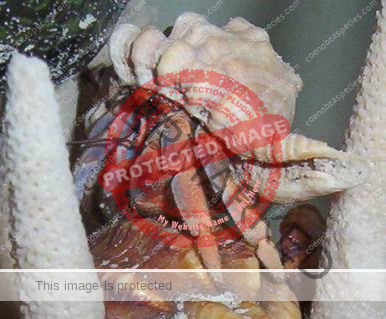
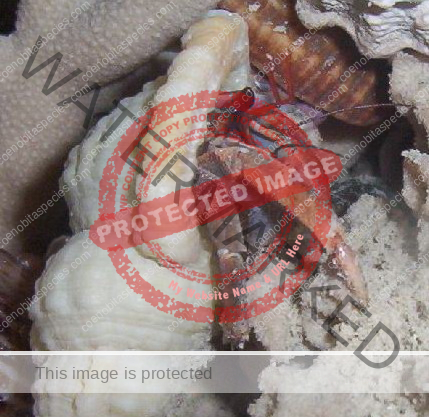
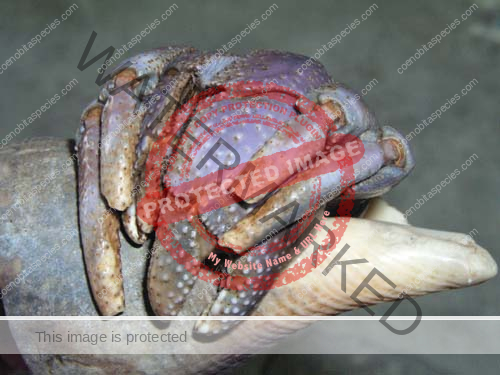
Characteristics: Entire body violet purple, but varying from light lavender to dark violet; dark patch of brown on outer lower surface of palm of left cheliped. [2] Juveniles are pale to orange or red with colors darkening as the crab ages. Eyes are elongated with the stalks being almost entirely black.
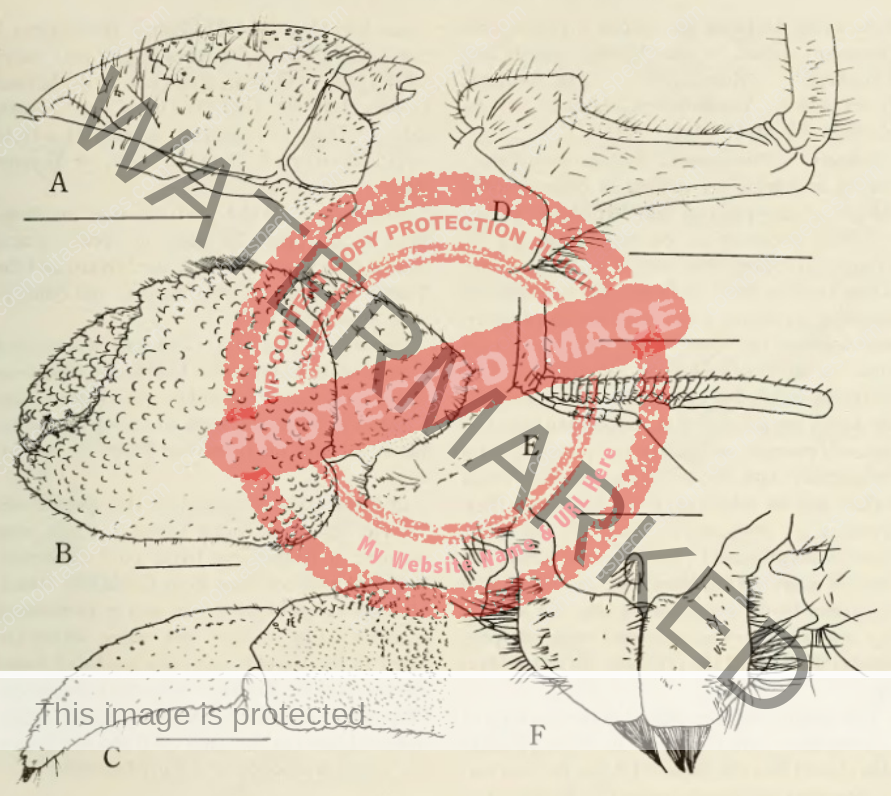
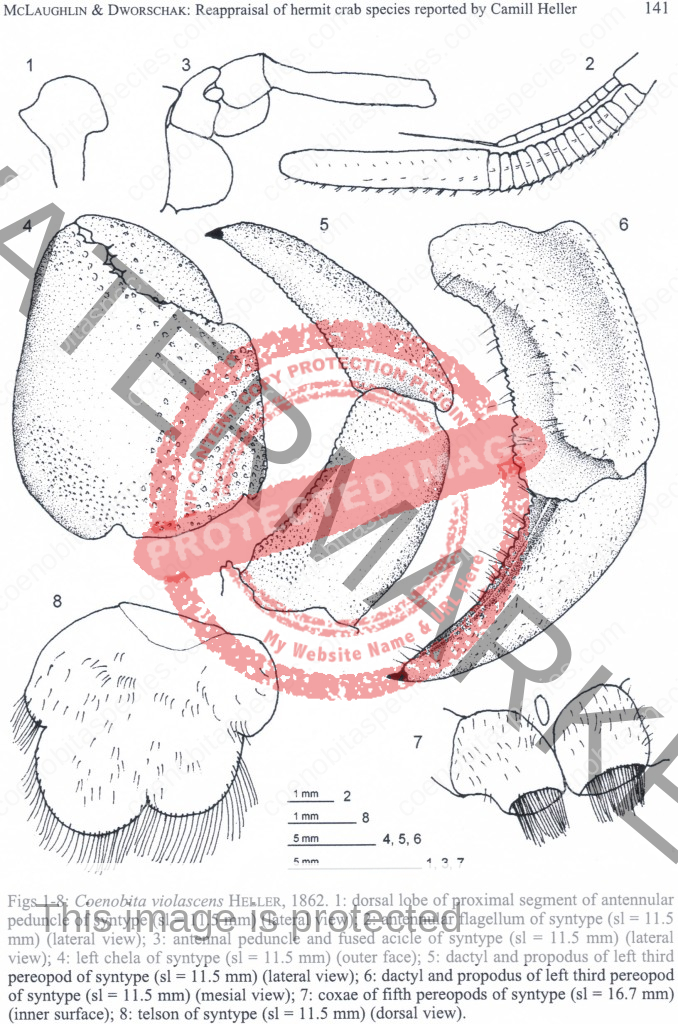
Dorsal surface of shield with scattered granules and punctations, lateral projections produced and each with terminal spinule; rostrum broadly triangular, not produced beyond level of lateral projections. Ocular peduncles compressed, reaching almost to mid-length of ultimate antennal peduncular segment; dorsal surfaces with numerous short setae; ocular acicles narrowly triangular, approximate. Antennular peduncles extremely long; basal peduncular segment (Fig. 1) with broad, roundly subrectangular dorsal lobe; lower ramus of flagellum (Fig. 2) approximately half length of upper. Antennal acicle (Fig. 3) fused with second peduncular segment. Each chela with tuft of dense setae on upper inner margin. Left cheliped without stridulating ridge on upper outer surface of palm (Fig. 4), but with numerous scattered granules on upper half of outer face, fewer on lower half and lower central region nearly smooth; lower margin of palm straight or slightly concave, lower proximal angle produced into lobe-like projection. Left third pereopod with lateral surface of propodus (Fig. 5) nearly smooth, separated from dorsal surface by well-marked longitudinal crest in anterior half, dorsomesial margin (Fig. 6) prominently projecting inward, upper half of mesial surface concave, median portion convex and with low ridge delineating concave ventromesial surface; dactyl broad, short, with dorsolateral margin (Fig. 5) distinctly angular, lateral face weakly convex, mesial surface (Fig. 6) with concave lower portion marked by distinct longitudinal ridge. Coxae of males (Fig. 7) subequal, approximate, both thick and short, each with dense tuft of setae; no sexual tubes developed; sternal protuberance relatively small. Telson (Fig. 8) with distinct incision separating anterior and posterior portions; terminal margins unarmed but with long dense setae over entire length. [3]
Body mass
Common Identifiers
- Eyes- elongated, eyestalks typically dark
- Shield – punctate, no setae or tubercles as in lila
- Claw- no stitch marks, setae on upper margin of palm,scattered tubercles on lower outer surface
- Color- juvenile orange/white/brown, becomes violet as adult, antenna are orange
- Chirp- yes
Behavior: usually found in mangrove forests along rivers and estuaries, (Needed: water preference, activity, how far they travel, tree climbing, breeding season)
Diet: omnivorous scavenger
Preferred shells: Terebridae (auger shells), whelks, fox shells
Violascens (Japan):
This is specific to C. violascens found in the Ryukyu Islands, Japan and is the first description of the larval releasing behavior and associated tree climbing in C. violascens. [4]
Note: “We did not capture any individuals of C. violascens and only observed and photographed their behaviors because all species of Coenobita have been protected in Japan since 1970 to promote their conservation.[4]
The larval release behavior of coenobitid crabs has generally been divided into three types with respect to their contact with seawater: (1) females immerse themselves in seawater and release larvae directly into the sea; (2) females do not immerse themselves in seawater and wait until they encounter a wave, and hatched larvae are then washed away b outgoing waves into the sea; (3) females do not immerse themselves in seawater and deposit their pre-hatching embryos onto emerged substrates when the waves move away from the shore. [4]
Coenobita violascens in this area is found only at the brackish mouth of rivers in which other coenobitid crabs are uncommon. This species is categorized as “near threatened” by the Japanese Association of Benthology because its abundance is lower than that of other species of Coenobita due to habitat loss as a result of over exploitations such as the modification of rivers on the islands. (Fujita 2012) [4]
Coenobita violascens took two consecutive days to complete spawning, interrupted during daylight hours. Crabs undertook vertical migrations by climbing the trees before releasing their larvae. During the nighttime low tide, ovigerous females dwelling in the coastal forest were observed approaching the mangrove trees along the intertidal riverbank below EHWS and climbing up to a height of 20-160 cm. No individuals were observed at the top of the tres. After climbing they rested on the trunk under the leaf canopy or under the root system to avoid direct sunlight during daytime. They descended to the roots the following night around high tide. All female crabs immersed themselves fully or partially in river water and swung back and forth while fastening themselves to the tree roots with the second and third pereiopods. Zoea larvae emerged from the shell aperture helped by the retracting movements of females. Larval releasing behavior lasted 45-160 seconds. [4]
Larval release by C. violascens on Iriomote Island took place only during the warm season, late July to early October, which corresponds to the larval release of some other semi-terrestrial crustaceans in the subtropical Ryukyu Islands. Breeding migration was observed from mid-June to late July for C. purpureus on Kikai island. Larval release by C. rugosus on Okinawa Island was observed from late June to late November, and from mid-June to early September in C. cavipes and C. purpureus. The breeding span of C. violascens and other semi-terrestrial crustaceans is limited to the nearly three-month warm season, with the exception of C. rugosus. Coenobita rugosus has a range that extends northward to the southern coast of mainland Japan, allowing for a longer reproductive period than congeners on the subtropical Ryukyu Islands. In contrast, C. violascens is a tropical species and the present study site was located at its northern marginal area, therefore a short reproductive period and a limited annual number of spawns per individual are expected. [4]
Compared with their congeneric nearest neighbor, C. cavipes, C. violascens has a considerably larger body size, shows a higher proportion of individuals protruding from shells and are unable to pull their left chela into the shell aperture. [4]
Coenobita violascens is adapted to the mangrove habitats, and releasing larvae on mangrove roots after climbing trees likely enhances the probability of as many larvae as possible reaching the sea compared with releasing larvae on the riverbank. [4]
The larval release of C. violascens (Japan) occurred around nighttime high tide during new moon periods, showing clear lunar and tidal rhythms. [4]
In contrast, the rhythm of larval release with regard to lunar and tidal phases is not clear for three congeners, C. rugosus, C. purpureus, and C. cavipes, all of which release larvae along the seacoast (Nakasone 2001). In these three species, larval release occurs mostly around high tide during both the full moon and new moon periods, but also occurs during other lunar phase periods; larval release occurs for 2 hours at spring tides, and become sporadic at neap tides. [4]
EHWS- Extreme High Water Spring tide [4]
Click any image to view full size:
C. violascens on Instagram
[insta-gallery id=”13″]
Coenobita violascens on Flickr
Photo credit:
The Crab Street Journal has been granted permission by these photographers to use their photo(s) on our site.
Nicholas
Felix Wang
References:
1. MCLAUGHLIN 1997,
2. NAKASONE 1988,
3. Heller 1862,
4. Larval release and associated tree climbing behavior of the land hermit crab Coenobita violascens Heller, 1862 Waturu Doi, Akira Mizutani, Hiroyoshi Kohno 2016
5. Larval growth, development and duration in terrestrial hermit crabs
Katsuyuki Hamasaki1,*, Saori Kato1, Yu Murakami1, Shigeki Dan2, Shuichi Kitada
6. Md. Sagir Ahmed, Tamim Afrin, Anindita Barua, New distributional record of Charybdis japonica, Coenobita violascens, Galene bispinosa, and Portunus reticulatus (Crustacea: Decapoda) from Bangladesh waters of the Bay of Bengal,
Regional Studies in Marine Science, Volume 44, 2021, 101785, ISSN 2352-4855,
https://doi.org/10.1016/j.rsma.2021.101785.





























































































.jpg)



































